|
Then the coefficients of the balanced chemical reaction can be used to convert to moles of another substance, which can then be converted to a mass.įor example, let us determine the number of grams of SO 3 that can be produced by the reaction of 45. You can use the third calculator (from page top) to calculate a dilution or preparation of a stock solution. Again, however, it is important to emphasize that before the balanced chemical reaction is used, the mass quantity must first be converted to moles. To get v/v percentage, multiply molarity by molar mass of the substance and divide by 10 times the mass density of the solution. Putting in a molecular formula of any type such as K2Cr2O7, CH3CH2COOH, KFeFe(CN)63, or Na2B4O7.10H20 will result in the molar mass and mass analyses. For this type of calculation, the molar masses of two different substances must be used-be sure to keep track of which is which. This mass is then divided by the molar mass of the compound and multiplied by 100: The molar mass of a chemical compound is. Atomic Mass shorts chemistry viral thediamondacademyyoutubeshorts youtube chemistry career. If you dont need the molecular formula, its not necessary to input the substances molar mass. If youd like to know the masses of C, H, and O of the sample, select Yes from the drop-down menu on the last row. Using the lower indices in the formula, the mass of each element in one mole of the compound is first calculated. The calculator will display your substances empirical formula, empirical mass, and molecular formula. 
It should be a trivial task now to extend the calculations to mass-mass calculations, in which we start with a mass of some substance and end with the mass of another substance in the chemical reaction. The percent composition of a compound can be easily determined from the chemical formula of the compound. 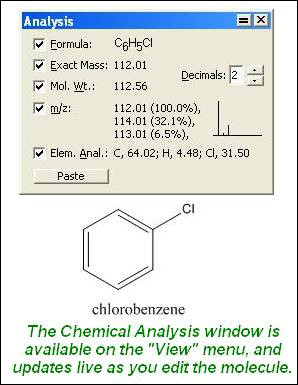 
In one line, 86.4 g K × 1 m o l K 39.09 g K × 1 m o l M g 2 m o l K × 24.31 g M g 1 m o l M g 26.87 g M g. You can use the mass percent calculator to determine your percentage ratio between the mass of a component and the total weight of the substance. In addition to the balanced chemical equation, we need the molar masses of K (39.09 g/mol) and Mg (24.31 g/mol). \nonumber \] Answerģ0.4 g (Note: here we go from a product to a reactant, showing that mole-mass problems can begin and end with any substance in the chemical equation.) Adding Chemistry Moles Calculator to your Wordpres website is fast and easy Find the page to which you want to add the calculator, go to edit mode. Using the chemical formula of a single isotope you can get the exact mass and the relative abundance percentage of.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
